Avelle™ Negative Pressure Wound Therapy Device
Choose Quantity
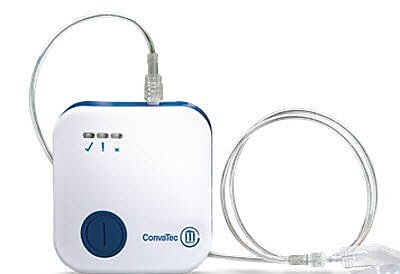
Avelle™ Negative Pressure Wound Therapy Device
The Avelle™ negative pressure wound therapy device is designed for advanced wound care applications where a negative pressure wound therapy system is required. This listing is built from the provided product title, MPN, and packaging data only.
Its single-unit format supports specialty wound care purchasing for clinics, treatment settings, and patient care programs that require advanced wound therapy equipment.
Key Features:
• Negative pressure wound therapy device
• Avelle™ brand
• Single-unit packaging
• Advanced wound care device format
• MPN: 422285
Benefits:
• Supports advanced wound care supply needs
• Single-unit format supports direct replacement and controlled purchasing
• Suitable for specialty wound care settings
• Professional-use device format supports structured care workflows
Specifications:
• Brand: Avelle™
• Product Type: Negative Pressure Wound Therapy Device
• Sterility: Not Verified
• Minimum Selling/Purchasing Unit: EA
• Minimum Package Qty: 1 EA
• Packaging: EA/1
• MPN: 422285
Application:
Designed for advanced wound care settings where a negative pressure wound therapy device is required.
• Wound care clinics
• Medical offices
• Physician offices
• Patient care support
• Specialty treatment settings
Warnings:
• For professional use according to product instructions
• Use only as directed for the intended wound care application
• Inspect product packaging before use
• Follow facility protocol and manufacturer guidance.






