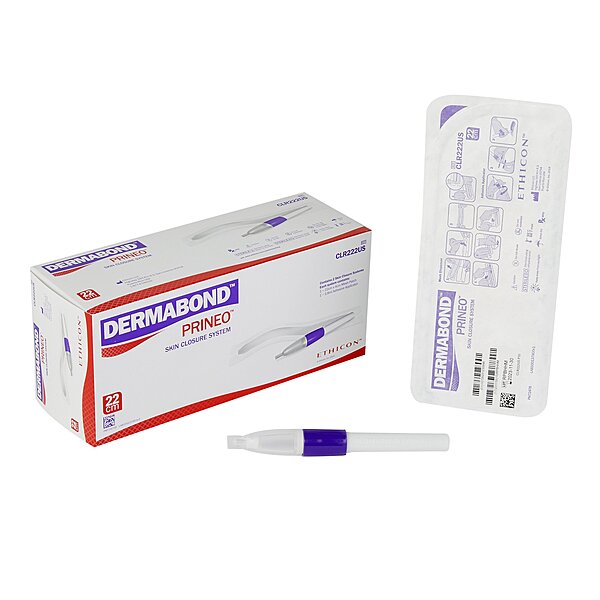
Dermabond® Prineo® Skin Closure System 2.9 mL Liquid Applicator Tip, 22 cm Mesh (BX/2EA)-BX
Dermabond® Prineo® Skin Closure System is a two-part liquid skin closure system featuring a 2.9 mL liquid applicator and 22 cm mesh component for skin closure use. MPN: CLR222US.
Choose Quantity
Dermabond® Prineo® Skin Closure System 2.9 mL Liquid Applicator Tip, 22 cm Mesh (BX/2EA)
Dermabond Prineo Skin Closure System is a two-part skin closure system that combines a liquid adhesive component with a flexible, self-adhesive polyester mesh. The system includes a 2.9 mL liquid applicator tip and a 22 cm mesh component designed to support skin closure workflow in professional care settings.
This product is supplied in a box of 2 systems for procedural supply standardization and facility stocking.
Key Features:
• Two-part skin closure system (liquid adhesive + self-adhesive polyester mesh)
• 2.9 mL liquid applicator tip
• 22 cm mesh component
• Liquid format
• Designed for skin closure system use
• Box of 2 systems
Benefits:
• Supports controlled application using an applicator tip
• Mesh component helps support skin approximation workflow
• Box quantity supports consistent facility stocking and replenishment
• Suitable for procedure-based supply programs
Specifications:
• Brand: Dermabond® Prineo®
• Product Type: Skin Closure System
• Type: Liquid
• Active Ingredients: 2-Octyl Monomer Adhesive
• Volume: 2.9 mL
• Tip Type: Applicator Tip
• Mesh Dimensions: 22 cm Mesh
• Buy American Act (BAA) Compliant: Yes
• Trade Agreement Act (TAA) Compliant: Yes
• Country of Origin: Unknown
• UNSPSC: 42312005
• Minimum Selling/Purchasing Unit: BX
• Minimum Package Qty: 2 Each
• Packaging: BX/2EA
• MPN: CLR222US
Application:
Skin closure systems are used in professional care environments as part of skin closure workflow.
Common applications include:
• Procedure-based clinical settings
• Facility wound-closure supply programs
• Situations requiring a liquid applicator and mesh-based skin closure approach
Warnings:
• For professional use
• Use according to product labeling and facility protocol
• Inspect packaging integrity prior to use
• Store and handle according to facility requirements







