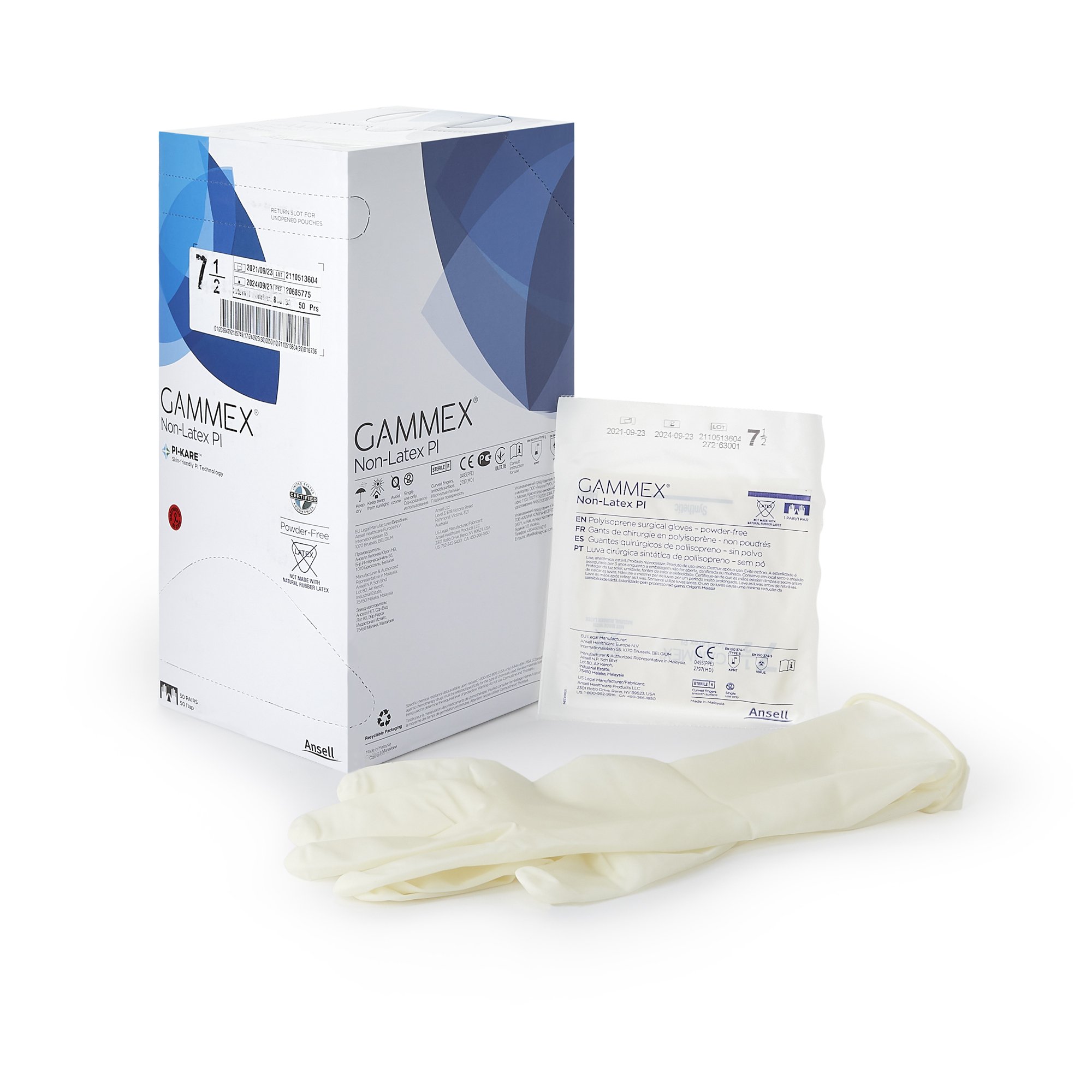

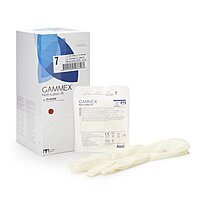
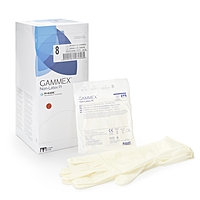
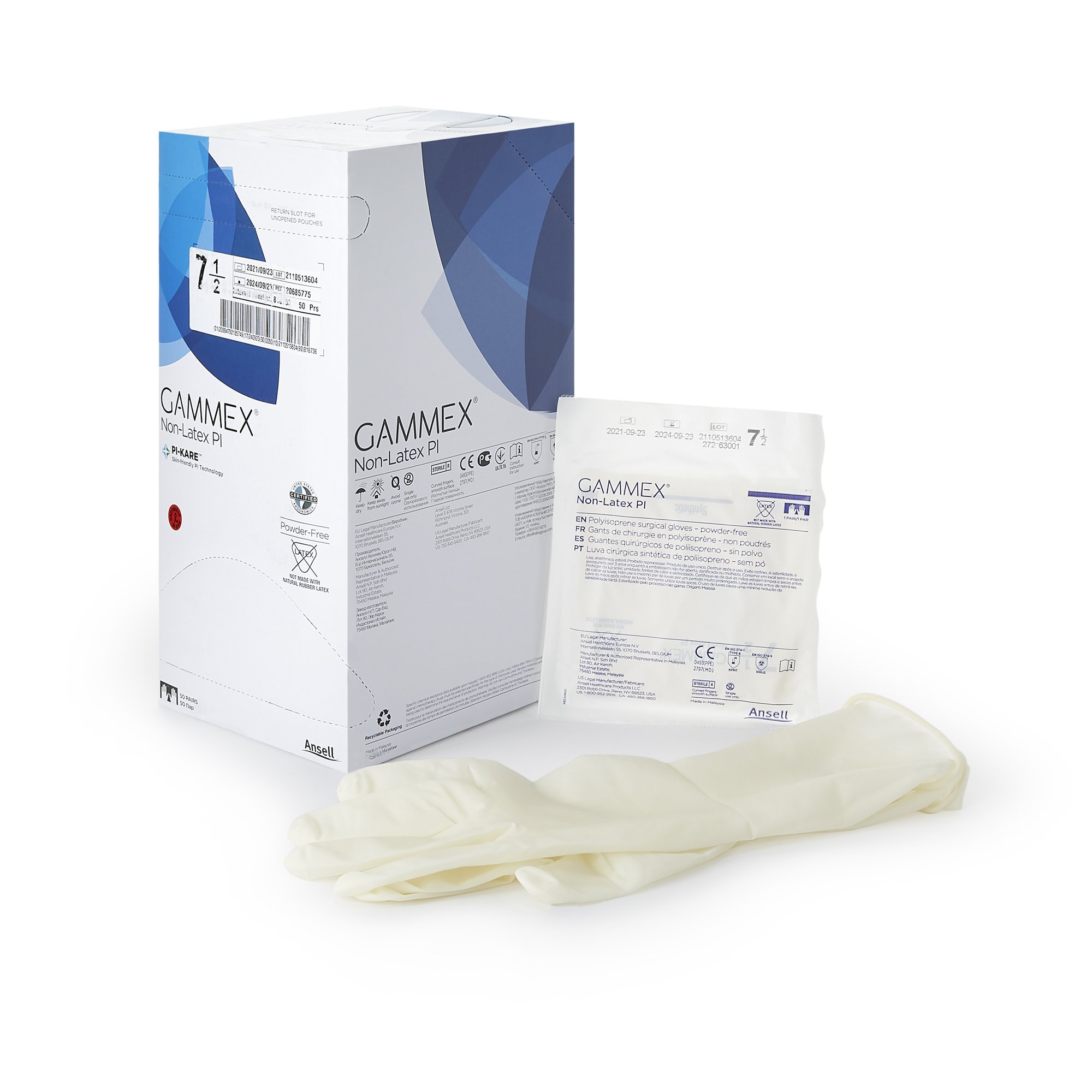
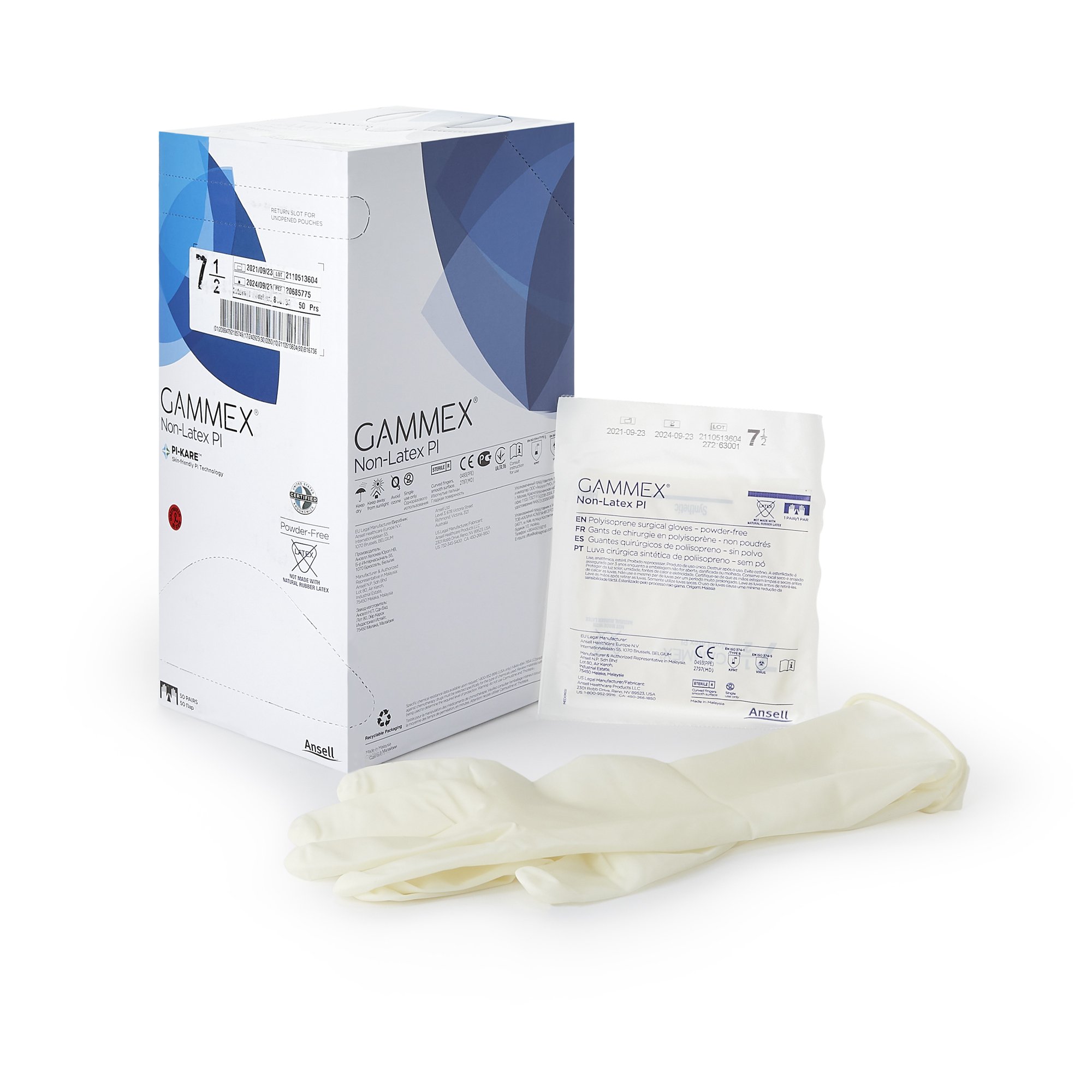
GAMMEX Non-Latex PI Sterile Polyisoprene Surgical Gloves Micro-Textured Chemo Tested (BX/50PR)
Choose Quantity
GAMMEX Non-Latex PI Sterile Polyisoprene Surgical Gloves Chemo Tested (BX/50PR)
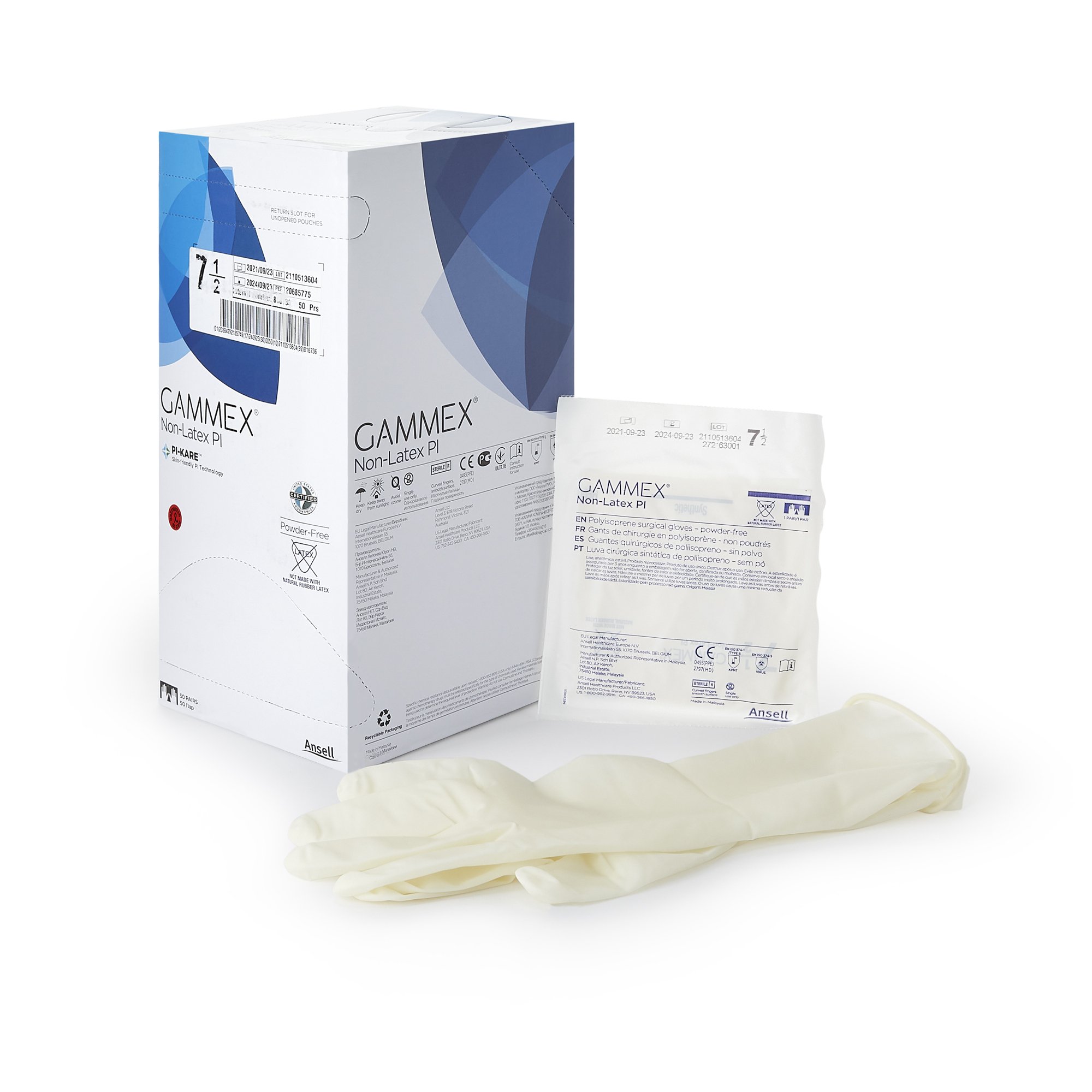
GAMMEX Non-Latex PI surgical gloves are sterile, powder-free surgical gloves made from synthetic polyisoprene for facilities and clinicians that prefer a non-latex glove option. These gloves feature a smooth surface with a micro-textured finish to support confident instrument handling and grip, along with a polymer coating on the interior to support donning in clinical workflows.
This is a variable product by size. Select the correct size (5.5 through 9) when ordering. Each size variant uses a different MPN, and each box contains 50 pairs (BX/50PR). Case packaging is 4 boxes per case (CS/4BX/50PR) for centralized purchasing and stocking.
Key Features:
•Sterile surgical glove made from synthetic polyisoprene
•Non-latex (polyisoprene) construction
•Micro-textured exterior for grip with smooth surface finish
•Interior polymer coating
•Beaded cuff
•Hand-specific design
•Chemo tested
•50 pairs per box (BX/50PR); 4 boxes per case (CS/4BX/50PR)
Benefits:
•Supports facilities seeking a non-latex sterile surgical glove option
•Micro-textured finish supports secure handling and instrument grip
•Hand-specific fit supports comfort and control during procedures
•Multiple sizes support standardization across surgical teams
•Box and case packaging supports flexible stocking and purchasing
Specifications:
•Brand: GAMMEX® Non-Latex PI
•Manufacturer: Ansell
•Product Type: Surgical Glove
•Material: Polyisoprene
•Color: White
•Glove Exterior: Micro-Textured (smooth surface with micro-textured finish)
•Glove Interior: With Polymer Coating
•Cuff Style: Beaded Cuff
•Glove Length: Standard Cuff Length
•Hand Compatibility: Hand Specific
•Length: 12 Inch
•Fingertip Thickness (Typical): 0.24 mm (9.4 mil)
•Protection Level: Chemo Tested
•Sterility: Sterile
•Quantity: 50 Pairs per Box
•HCPCS: A4930
•UNSPSC: 42132205
•Country of Origin: Unknown
•TAA Compliant: No
•BAA Compliant: No
•Minimum Selling/Purchasing Unit: BX
•Minimum Package Qty: 50 PR per BX
•Packaging (Factual): 50PR/BX; 4BX/CS (CS/4BX/50PR)
•MPN Varies by Variant
•Size 5.5 → 20685755
•Size 6 → 20685760
•Size 6.5 → 20685765
•Size 7 → 20685770
•Size 7.5 → 20685775
•Size 8 → 20685780
•Size 8.5 → 20685785
•Size 9 → 20685790
Application:
Sterile surgical gloves intended for use in surgical and sterile procedure environments.
•Select the correct size variant for the user
•Use according to facility protocol for sterile glove donning and intraoperative changes
Warnings:
•Single-use only; do not reuse
•Sterile product; do not use if packaging is damaged or opened prior to use
•Follow facility policy for selection and use when handling chemotherapy agents and other chemicals
•Discontinue use if irritation occurs







