




McKesson IV Start Kit Sterile (EA)
Choose Quantity
McKesson IV Start Kit Sterile (EA)
McKesson IV Start Kit is a sterile single-use kit designed for IV start procedures in professional care settings. The kit is assembled to support organized vascular access setup and includes commonly used components for routine IV start workflow.
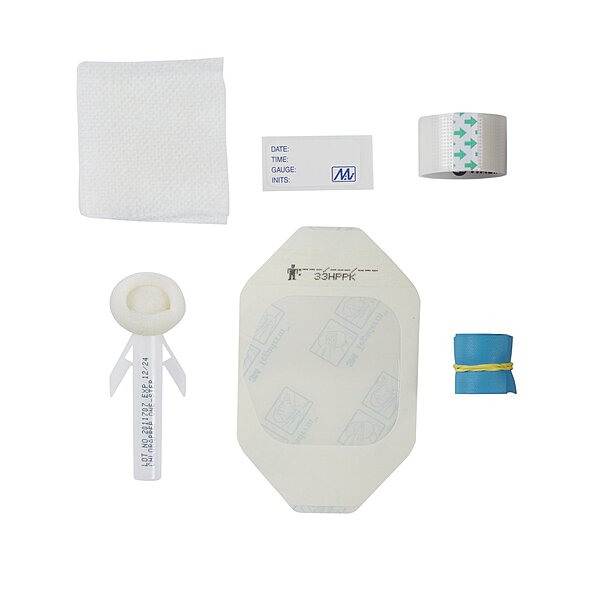
Contents include 1 tourniquet, 1 ChloraPrep® 1 mL applicator with DFU, 2 non-woven gauze pads, 1 Tegaderm™ dressing, 1 roll of Transpore™ tape, and 1 IV change label. The kit is not made with natural rubber latex.
This product is suitable for hospitals, outpatient infusion centers, urgent care settings, physician offices, and clinics where sterile IV start supplies are maintained as part of regular procedural inventory.
Key Features:
• Sterile IV start kit
• Single-use format
• Includes Tegaderm™ dressing
• Includes ChloraPrep® 1 mL applicator
• Includes tourniquet, gauze, tape, and IV change label
• Not made with natural rubber latex
• Case contains 50 kits
Benefits:
• Supports organized IV start workflow
• Single-use sterile format helps simplify procedural setup
• Includes commonly used components in one kit
• Suitable for routine vascular access supply programs
Specifications:
• Brand: McKesson
• Product Type: IV Start Kit
• Application: IV Start Kit
• Sterility: Sterile
• Usage: Single Use
• Contents: 1 Tourniquet, 1 ChloraPrep® 1 mL Applicator with DFU, 2 Non-Woven Gauze 2 X 2 Inch (4 Ply), 1 Tegaderm™ Dressing 2.37 X 2.7 Inch, 1 Roll of Transpore™ Tape, 1 IV Change Label
• Latex-Free: Not Made with Natural Rubber Latex
• Country of Origin: China
• UNSPSC: 42221507
• Minimum Selling/Purchasing Unit: EA or CS
• Minimum Package Qty: 1 Each
• Packaging: CS/50EA; EA/1
• MPN: 25-5818
Application:
McKesson IV Start Kit is used in professional care environments for IV start procedures and vascular access setup.
Common applications include:
• Hospitals and outpatient infusion settings
• Physician offices and clinics
• Urgent care environments
• General IV therapy supply programs
Use according to product labeling and facility protocol.
Warnings:
• For professional use
• Sterile unless package is opened or damaged
• Single-use only
• Inspect packaging before use






