


McKesson Suture Removal Kit Sterile Single Use Latex Free
Choose Quantity
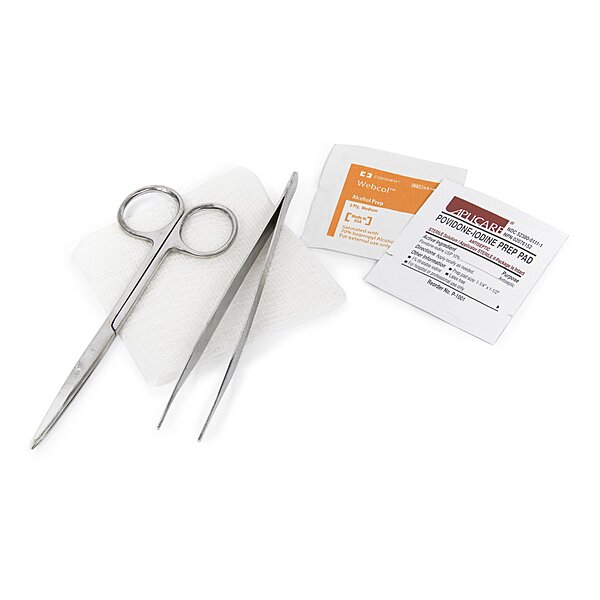
The McKesson Suture Removal Kit (MPN 25-5723) is a sterile, single-use kit designed to support routine suture removal workflows in clinical settings. It includes essential instruments and prep items packaged together to help standardize setup and support efficient procedure flow.
Kit Contents
This kit contains the following components:
•1 - Metal Iris Scissors with point protector, 4-1/2 inch
•1 - Metal Adson forceps, serrated, 4-3/4 inch
•1 - Gauze sponge, 3" x 3" (8-ply)
•1 - Alcohol prep pad
•1 - PVP prep pad
Sterility and Use
The kit is listed as sterile and intended for single use. It is also listed as not made with natural rubber latex, supporting latex-sensitive environments.
Packaging and Inventory Handling
This item is packaged as 50 kits per case (CS/50EA).
For your Zoho setup: the minimum selling unit is EA (1 kit), and the master unit is CS (50 kits) (both should be priced and active).
Key Features:
•McKesson suture removal kit with metal forceps
•Sterile
•Single use
•Not made with natural rubber latex
•Packaged 50 kits per case
Benefits:
•Standardizes suture removal setup with a consistent kit format
•Includes key instruments and prep items in one kit
•Supports latex-sensitive environments (latex-free indicator)
•Case quantity supports efficient stocking and replenishment
Specifications:
•Brand: McKesson
•Manufacturer: McKesson Brand
•MPN: 25-5723
•Application: Suture Removal Kit
•Sterility: Sterile
•Usage: Single Use
•Latex Free Indicator: Not Made with Natural Rubber Latex
•Contents: Metal iris scissors w/ point protector (4-1/2"), metal Adson forceps serrated (4-3/4"), 3" x 3" gauze sponge (8-ply), alcohol prep pad, PVP prep pad
•Country of Origin: China
•HCPCS: A4649
•UNSPSC: 42312207
•Packaging: 50 kits per case (CS/50EA)
Application:
Use as a prepared suture removal kit to support removal procedures per facility protocol.
•Open using appropriate aseptic technique
•Use included instruments and prep items per clinical policy
•Dispose after use according to facility guidelines
Warnings:
•Single-use only; do not reuse or reprocess.
•Do not use if sterile package is opened, damaged, or compromised.
•Follow facility protocol for disposal of used instruments and materials.






