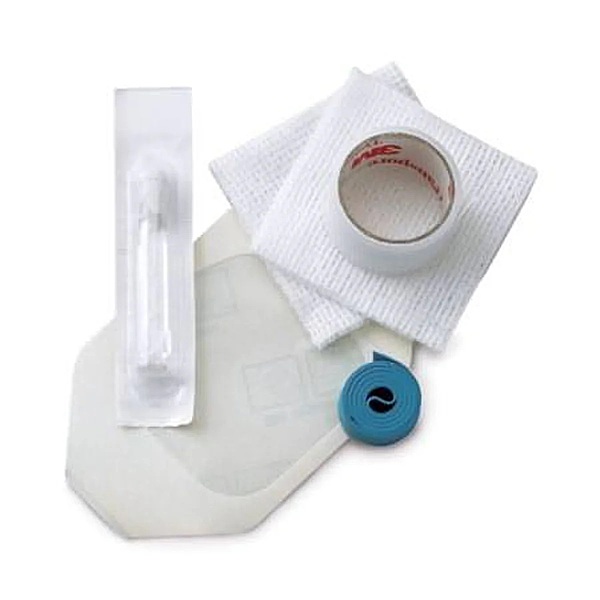
Medical Action IV Start Kit with Chloraprep, Dressing, Gauze, Tape & Tourniquet - CS
Choose Quantity
The Medical Action IV Start Kit (MPN 71952) is a convenient, standardized kit designed to support peripheral IV start preparation by grouping commonly used components together in a single kit. This format helps streamline supply staging for clinical teams by keeping essential items organized and ready for use according to facility protocol.
Kit Contents
Each IV start kit includes the following components:
• (1) 2 3/8" x 2 3/4" dressing
• (2) 2" x 2" 4-ply gauze sponges
• (1) 1 mL Chloraprep applicator
• (1) 0.75" x 18" transparent tape
• (1) 0.75" x 18" rolled blue tourniquet
Packaging and Inventory Handling
This product is supplied as a case of 100 kits (CS/100EA). This listing is intended to be sold by the case as the minimum selling/purchasing unit, supporting bulk replenishment for clinics, urgent care, ambulatory centers, and procedure rooms.
Use Considerations
An IV start kit helps ensure that key prep items are available in one place at the time of care. Always follow your facility’s IV start policy and manufacturer labeling for use, storage, and disposal practices.
Key Features:
•IV start kit with multiple components in one kit
•Includes dressing, gauze, 1 mL Chloraprep applicator, transparent tape, and rolled blue tourniquet
•Standardized kit format for supply staging
•Packaged 100 kits per case (CS/100EA)
Benefits:
•Helps streamline IV start setup by consolidating key items
•Supports consistent stocking and replenishment by case quantity
•Reduces time spent gathering individual components
Specifications:
•Brand: Medical Action Industries
•MPN: 71952
•Product Type: IV Start Kit
•Kit Contents: (1) 2 3/8" x 2 3/4" dressing; (2) 2" x 2" 4-ply gauze; (1) 1 mL Chloraprep; (1) 0.75" x 18" transparent tape; (1) 0.75" x 18" rolled blue tourniquet
•Packaging: CS/100EA (100 kits per case)
Application:
Use for staging commonly needed supplies for peripheral IV start preparation per facility protocol.
•Open using appropriate aseptic technique per policy
•Dispose of used components per facility and local requirements
Warnings:
•Use according to manufacturer labeling and facility policy.
•Do not use if packaging is damaged or compromised.
•Single-use components—do not reuse.






